Diabetes is one of the most widespread chronic diseases of our time. Globally, an estimated 537 million adults were living with diabetes in 2021, and that number is projected to climb to 783 million by 2045, according to the IDF Diabetes Atlas.
Behind these numbers are real people navigating daily challenges, from monitoring blood sugar to managing medications and making sense of conflicting dietary advice.
The vast majority of these cases are type 2 diabetes, a condition that often develops silently, over years, before it is detected. By the time a diagnosis is made, many people already have some degree of organ damage.
Yet type 2 diabetes is not inevitable, and for many it is not even permanent. With the right understanding, it can be delayed, managed effectively, and in some cases put into remission.
This guide covers the full picture: what diabetes actually is, how type 2 differs from other forms, how it is diagnosed, what complications can follow if left unmanaged, what treatment options exist today, and how diet and lifestyle remain the most powerful tools available.
It also explores an exciting area of current science, the connection between gut health and blood sugar control.
Understanding Diabetes: The Different Types
Diabetes mellitus is not a single disease. It is a group of metabolic conditions that share one defining feature: chronically elevated blood glucose levels. This happens because the body cannot produce enough insulin, cannot use insulin properly, or both.
Insulin is a hormone produced by beta cells in the pancreas. Its job is straightforward but vital. When you eat carbohydrates, they break down into glucose and enter your bloodstream.
Insulin acts as a signal that tells your body’s cells to open up and absorb that glucose for energy. Without sufficient insulin or without cells that respond to it, glucose accumulates in the blood. Over time, this causes widespread damage.
Type 1 Diabetes
Type 1 diabetes is an autoimmune condition. The immune system mistakenly destroys the beta cells in the pancreas, leaving the body unable to produce insulin. People with type 1 diabetes must take insulin every day to survive. It typically appears in childhood or young adulthood but can develop at any age. It accounts for roughly 5 to 10 percent of all diabetes cases worldwide.
Type 2 Diabetes
Type 2 diabetes is by far the most common form, representing approximately 90 to 95 percent of all diagnoses.
The main difference between type 1 and type 2 diabetes is that in type 2, the body still produces some insulin. The core problem is that cells have become resistant to insulin’s signals, a condition called insulin resistance. The pancreas tries to compensate by producing more insulin, but over time, it cannot keep up, and blood glucose levels rise persistently.
Type 2 diabetes develops gradually, often over a decade or more, and is strongly linked to lifestyle factors including physical inactivity, poor diet, and excess body weight. Genetic predisposition also plays a meaningful role.
Gestational Diabetes
Gestational diabetes develops during pregnancy in women who did not previously have diabetes. It typically resolves after delivery, but both the mother and the child carry a significantly elevated risk of developing type 2 diabetes later in life.
Less Common Forms
Maturity-Onset Diabetes of the Young (MODY) is caused by single-gene mutations that affect insulin production and is often mistaken for type 1 or type 2. Latent Autoimmune Diabetes in Adults (LADA) shares features of both autoimmune and insulin-resistant diabetes and is frequently misclassified as type 2.
Type 2 Diabetes: What Happens Inside the Body
Understanding the biology of type 2 diabetes helps clarify why the condition progresses the way it does and why early intervention changes the trajectory so substantially.
In a healthy body, after a meal, blood glucose rises, and the pancreas responds quickly with insulin. Cells absorb the glucose, blood sugar normalises, and insulin levels fall. This process is efficient and precisely regulated.
In type 2 diabetes, this system breaks down in two stages:
In the first stage, cells, particularly in the muscle, liver, and fat tissue, become less responsive to insulin. The pancreas senses rising blood glucose and pumps out more insulin to compensate. For years, sometimes decades, blood glucose levels remain near normal because the pancreas is working overtime.
In the second stage, the beta cells, under relentless demand, begin to deteriorate. Insulin output declines. The compensatory mechanism fails, and blood glucose starts rising in a sustained, chronic way. This persistent hyperglycemia is what causes the damage to blood vessels, nerves, and organs that defines the complications of diabetes.
Take a look at the table for clearance:
| Stage 1: Compensation | Stage 2: Decline |
| Cells Become resistant to insulin signals | Beta cells Deteriorate under relentless demand |
| Pancreas Pumps out more insulin to compensate | Insulin output Declines as the pancreas wears out |
| Blood glucose Remains near normal (system overworks) | Blood glucose Rises in a sustained, chronic way |
| Timeline Can last years or even decades | Outcome Persistent hyperglycemia, organ damage |
Excess abdominal fat is a central driver of insulin resistance. Visceral fat, the fat stored around internal organs, releases inflammatory signals and free fatty acids that interfere directly with insulin signaling.
Inflammation, oxidative stress, and mitochondrial dysfunction all contribute to the progressive decline of beta-cell function.
How Type 2 Diabetes Is Diagnosed
One of the most important challenges with type 2 diabetes is that it is often asymptomatic in its early stages. Many people live with elevated blood glucose for years without knowing it. Type 2 diabetes symptoms, when they do appear, can include,
- Increased thirst
- Frequent urination
- Unexplained fatigue
- Blurred vision
- Slow wound healing
Diagnosis is based on blood tests. The criteria used by the American Diabetes Association and mirrored in international guidelines are as follows.
Fasting Plasma Glucose (FPG): Blood is drawn after at least eight hours of fasting. A reading of 126 mg/dL (7.0 mmol/L) or higher on two separate occasions confirms a diabetes diagnosis. A result between 100 and 125 mg/dL indicates impaired fasting glucose, commonly called prediabetes.
HbA1c (Glycated Haemoglobin): This test reflects average blood glucose over the previous two to three months by measuring how much glucose has attached to haemoglobin in red blood cells. An HbA1c of 6.5 percent or higher on two occasions confirms diabetes. A reading between 5.7 and 6.4 percent indicates prediabetes.
Oral Glucose Tolerance Test (OGTT): The patient drinks a standardised glucose solution, and blood sugar is measured two hours later. A result of 200 mg/dL or above confirms diabetes.
Random Plasma Glucose: A blood glucose of 200 mg/dL or more at any time, combined with classic symptoms, is diagnostic.
The prediabetes window is critically important. It is a reversible state. Without lifestyle changes, a substantial proportion of people with prediabetes will progress to type 2 diabetes within five to ten years.
This type 2 diabetes chart depicts diagnosis tests more clearly:
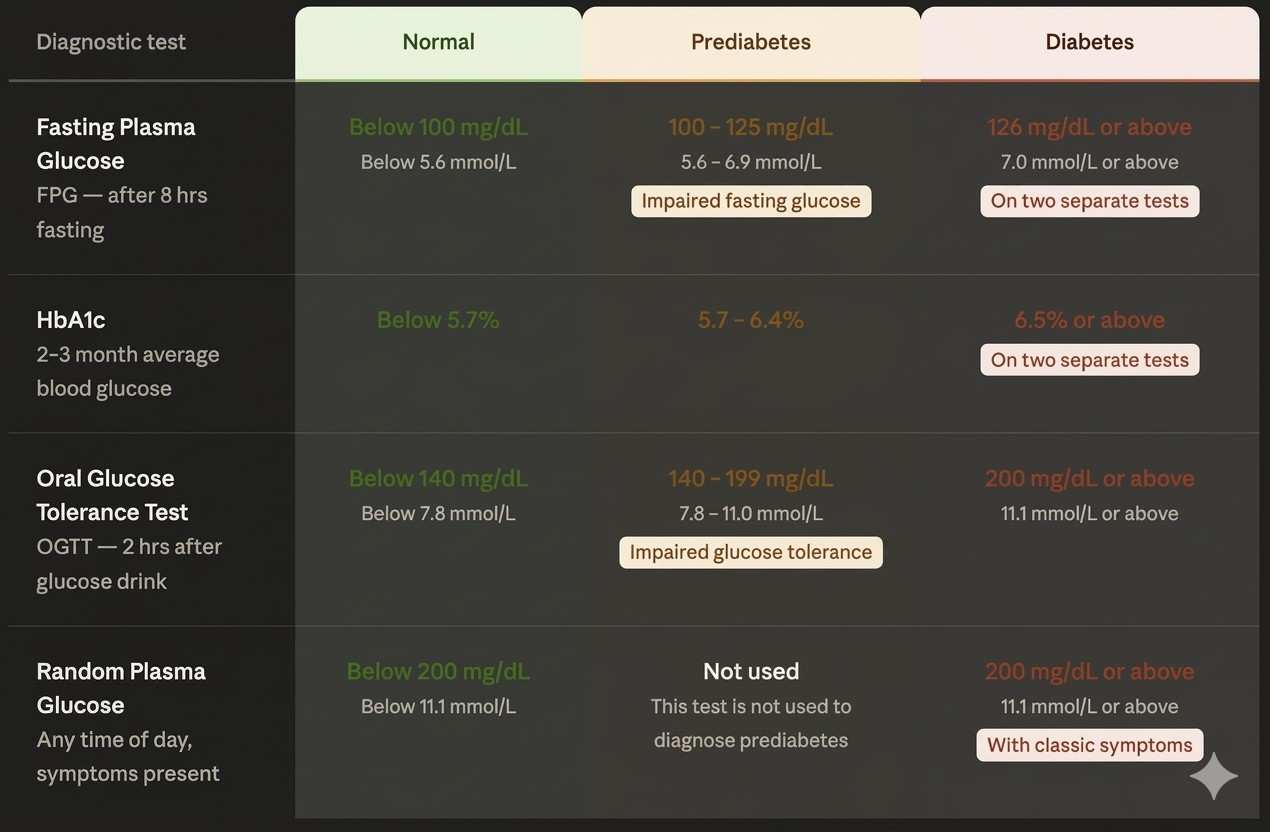
With appropriate intervention, this transition can be substantially delayed or prevented entirely. The case for early detection is compelling precisely because the tools for prevention are already well understood.
Complications of Type 2 Diabetes
Poorly managed type 2 diabetes does not simply mean elevated numbers on a blood test. Over the years, chronically high blood glucose degrades blood vessels and nerves throughout the body, leading to a cascade of serious, often debilitating complications.
Cardiovascular Disease
Cardiovascular disease is the leading cause of death in people with type 2 diabetes. Diabetes accelerates atherosclerosis, the accumulation of fatty plaques within artery walls, raising the risk of heart attack and stroke substantially. This elevated cardiovascular risk is the reason why newer diabetes medications are now specifically evaluated for their effects on heart outcomes, not just blood sugar numbers.
Diabetic Nephropathy
The kidneys rely on a dense network of tiny blood vessels to filter waste from the blood. Chronic hyperglycemia damages these vessels progressively, impairing kidney function over time. Diabetic nephropathy is one of the most common causes of end-stage kidney disease and dialysis dependency worldwide.
Diabetic Retinopathy
The blood vessels of the retina are among the most sensitive in the body. Sustained high blood glucose causes them to leak, swell, or grow abnormally. Diabetic retinopathy is a leading cause of blindness in working-age adults. Crucially, it often causes no symptoms until it is advanced, making regular dilated eye exams non-negotiable for anyone with diabetes.
Diabetic Neuropathy
Nerve damage from chronic hyperglycemia typically begins in the longest nerve fibres, which means the feet and legs are affected first. Symptoms include tingling, numbness, burning pain, or a complete loss of sensation. Loss of protective sensation in the feet dramatically increases the risk of unnoticed wounds, infections, and, in severe cases, amputation.
Increased Infection Susceptibility
High blood glucose impairs immune cell function and slows the healing of wounds and tissues. People with diabetes are more vulnerable to bacterial and fungal infections, and infections that would be minor in a non-diabetic person can escalate rapidly.
The encouraging reality is that the majority of these complications can be prevented or substantially delayed with consistent blood sugar management, along with attention to blood pressure, cholesterol, and lifestyle.
The Treatment Landscape
Diabetes treatment has evolved significantly over the past fifteen years. The goal of treatment is not only to lower blood glucose but to protect the heart, kidneys, and nervous system, and to improve quality of life.
Lifestyle Intervention: The Non-Negotiable Foundation
Lifestyle management or change is not a soft recommendation. It is the most evidence-backed intervention available.
The landmark Diabetes Prevention Program (DPP), a large randomised controlled trial published in The New England Journal of Medicine, found that intensive lifestyle intervention reduced the progression from prediabetes to type 2 diabetes by 58 percent. That outperformed metformin, which reduced incidence by 31 percent.
Metformin
Metformin remains the first-line medication for type 2 diabetes in most international guidelines. It works primarily by reducing glucose output from the liver and improving insulin sensitivity in peripheral tissues.
It is inexpensive, well-tolerated, and has a decades-long safety record. Newer research suggests metformin also exerts metabolic effects through the gut, which is explored further below.
SGLT2 Inhibitors
Sodium-glucose co-transporter 2 (SGLT2) inhibitors, including empagliflozin and dapagliflozin, work by blocking glucose reabsorption in the kidneys, causing excess glucose to be excreted in urine.
Beyond blood sugar control, this drug class has shown substantial benefits for cardiovascular and kidney health. The EMPA-REG OUTCOME trial, published in The New England Journal of Medicine, demonstrated that empagliflozin significantly reduced cardiovascular death and hospitalisation for heart failure in people with type 2 diabetes and established cardiovascular disease.
GLP-1 Receptor Agonists
GLP-1 receptor agonists, including semaglutide and liraglutide, mimic the effects of a naturally occurring gut hormone. They stimulate insulin release in response to meals, suppress glucagon (a hormone that raises blood glucose), slow gastric emptying, and reduce appetite.
The result is meaningful blood sugar reduction alongside significant weight loss, an uncommon combination in diabetes pharmacology. The LEADER trial, published in The New England Journal of Medicine, showed that liraglutide reduced major cardiovascular events in people with type 2 diabetes at high cardiovascular risk.
DPP-4 Inhibitors
Dipeptidyl peptidase-4 (DPP-4) inhibitors, such as sitagliptin and saxagliptin, extend the action of naturally occurring incretin hormones, which stimulate insulin secretion and suppress glucagon in a glucose-dependent manner. They have a low risk of causing hypoglycemia and are generally well-tolerated.
Insulin Therapy
When other medications are insufficient to control blood glucose, or when blood glucose is very high at diagnosis, insulin therapy is initiated. Formulations range from long-acting basal insulin given once daily to rapid-acting mealtime insulin doses.
Modern insulin delivery systems, including insulin pens and continuous glucose monitors, have made insulin therapy more precise and manageable than earlier generations of treatment.
The Possibility of Remission
Diabetes remission, defined as sustained normal blood glucose without medication, is a realistic goal for some people. The DiRECT trial, published in The Lancet, demonstrated that intensive dietary weight management could achieve remission of type 2 diabetes in approximately 46 percent of participants at one year. Those who lost 15 kg or more achieved the highest rates of remission.
This finding has reshaped how clinicians and patients think about the condition: not as irreversible, but as highly responsive to sustained lifestyle change.
Diet and Nutrition
What you eat is one of the most direct levers for influencing blood glucose, insulin sensitivity, body weight, and gut health. There is no single correct diet for type 2 diabetes, but the principles of what works are well-established.
The Mediterranean Diet
The Mediterranean dietary pattern consistently shows the strongest evidence for metabolic health. It emphasises vegetables, fruits, whole grains, legumes, nuts, and olive oil, with limited red meat and processed food.
The large PREDIMED randomised controlled trial, published in The New England Journal of Medicine, found that a Mediterranean diet supplemented with extra virgin olive oil or nuts significantly reduced major cardiovascular events in high-risk individuals, including those with diabetes.
Low-Carbohydrate Diets
Reducing carbohydrate intake lowers postprandial glucose spikes and reduces the insulin demand on an already stressed pancreas. Low-carbohydrate approaches have been shown in multiple clinical trials to improve HbA1c and reduce medication requirements, particularly in the short to medium term.
The degree of restriction, from moderate carbohydrate reduction to very low-carbohydrate ketogenic approaches, can be personalised to individual preference and tolerance.
The Role of Dietary Fibre
Dietary fibre is arguably the single most important dietary variable for blood sugar and gut health combined. Soluble fibre slows the absorption of glucose, reduces postprandial glucose peaks, and feeds beneficial gut bacteria.
A major meta-analysis published in The Lancet found that higher dietary fibre intake was associated with significantly lower risk of developing type 2 diabetes, as well as improved blood glucose control and reduced all-cause mortality in people already diagnosed.
High-fibre foods include:
- Oats
- Barley
- Lentils
- Chickpeas
- Flaxseeds
- Vegetables
- Whole fruits
Aiming for 25 to 35 grams of fibre per day is a reasonable target for most adults.
Foods to Prioritise and Foods to Limit
Prioritise non-starchy vegetables, legumes, whole grains, lean proteins, foods rich in anti-inflammatory omega-3 fatty acids, nuts and seeds, and olive oil.
Limit refined carbohydrates, white bread, sugary beverages, ultra-processed snack foods, fried foods, and high quantities of red and processed meats.
Consistent meal timing and appropriate portion sizes matter as much as food selection. Large, carbohydrate-heavy meals create sharp glucose spikes. Eating at regular intervals throughout the day and keeping meals balanced helps maintain more stable blood sugar levels.
Lifestyle Factors Beyond Diet
Diet works best when it is part of a broader lifestyle approach. Several other daily habits have a meaningful, evidence-backed impact on blood sugar regulation.
Physical Activity
Exercise improves insulin sensitivity directly, lowering blood glucose and reducing cardiovascular risk at the same time. Both aerobic exercise, such as walking, cycling, or swimming, and resistance training, such as weight lifting or bodyweight exercises, benefit blood sugar control.
Research published in Annals of Internal Medicine found that combining aerobic and resistance training was more effective at improving HbA1c than either type of exercise performed alone.
Current guidelines recommend at least 150 minutes of moderate-intensity aerobic activity per week, along with two or more sessions of resistance training. Even simple habits, like a 10-minute walk after meals, meaningfully reduce postprandial blood glucose levels.
Sleep Quality
Sleep is a metabolic necessity, not a luxury. Short sleep duration and poor sleep quality are both independently associated with increased insulin resistance and elevated stress hormone levels.
Chronic sleep deprivation disrupts glucose metabolism at a cellular level, making blood sugar harder to control. Most adults require seven to nine hours of quality sleep per night. For people with diabetes, prioritising sleep is not optional.
Stress Management
Psychological stress triggers the release of cortisol and adrenaline, hormones that signal the liver to release glucose into the bloodstream. Chronic stress, therefore, directly worsens blood sugar control, even without any change in diet or physical activity.
Evidence supports the role of mindfulness-based practices, breathing exercises, yoga, and social connectedness in reducing the physiological burden of stress. A systematic review published in Diabetes Research and Clinical Practice found that mindfulness-based interventions were associated with modest improvements in HbA1c and psychological well-being in people with type 2 diabetes.
Smoking Cessation
Nicotine impairs insulin secretion and promotes insulin resistance. Smoking also compounds the already elevated cardiovascular risk associated with type 2 diabetes. Stopping smoking is one of the highest-impact changes a person with diabetes can make for long-term health outcomes.
Weight Management
Even modest weight loss, five to ten percent of total body weight, produces clinically meaningful improvements in insulin sensitivity, blood pressure, and lipid levels. More significant weight loss substantially increases the possibility of diabetes remission, as demonstrated in the DiRECT trial.
Weight management does not require perfection. Gradual, sustainable change consistently outperforms extreme short-term approaches.
The Gut Microbiome and Type 2 Diabetes
Perhaps the most compelling recent development in diabetes research is the recognition that the trillions of microorganisms living in the human gut play a direct role in blood glucose regulation, insulin sensitivity, and systemic inflammation.
This field is moving quickly, and the evidence is increasingly hard to ignore.
What the Research Shows
A landmark 2012 study by Qin and colleagues, published in Nature, conducted a metagenome-wide association study in Chinese adults and found that people with type 2 diabetes had measurable differences in their gut microbial communities compared to those without it. Critically, those with diabetes showed a reduction in butyrate-producing bacteria, a finding that has significant implications for metabolic health.
A follow-up study by Karlsson and colleagues, published in Nature in 2013, replicated similar findings in a European cohort of women. Specific gut bacteria were identified whose relative abundance correlated with whether an individual had normal, impaired, or diabetic glucose control.
These findings established a clear association between gut dysbiosis, an imbalance in gut microbial communities, and type 2 diabetes.
Short-Chain Fatty Acids: A Key Mechanism
The gut’s connection to blood sugar is not just correlational. There are well-understood biological mechanisms at work, and short-chain fatty acids (SCFAs) are central to them.
When gut bacteria ferment dietary fibre, they produce SCFAs, primarily butyrate, propionate, and acetate. Butyrate is the primary fuel source for colonocytes, the cells lining the large intestine, and it plays a critical role in maintaining the integrity of the gut lining. It also has anti-inflammatory properties and appears to improve insulin sensitivity in peripheral tissues.
In people with type 2 diabetes, bacteria that produce butyrate, particularly Roseburia intestinalis and Faecalibacterium prausnitzii, are often depleted. This reduction in butyrate production may contribute to increased intestinal permeability, a state in which the gut lining becomes more permeable than it should be.
Bacterial fragments and other inflammatory molecules can then pass into the bloodstream, triggering chronic low-grade systemic inflammation, which in turn worsens insulin resistance.
Akkermansia Muciniphila
One specific bacterium attracting considerable research attention is Akkermansia muciniphila. This species colonises the mucus layer of the gut lining and plays a role in maintaining barrier integrity and metabolic health.
Lower levels of A. muciniphila have been consistently observed in people with obesity and type 2 diabetes.
A clinical study by Depommier and colleagues, published in Nature Medicine in 2019, found that oral supplementation with pasteurised A. muciniphila improved insulin sensitivity, reduced fasting blood glucose, and lowered relevant blood lipids in overweight and obese adults with metabolic syndrome.
While further clinical trials are needed, this finding positions A. muciniphila as one of the most promising therapeutic targets in metabolic disease research.
Metformin Works Partly Through the Gut
One of the more surprising findings in recent diabetes pharmacology is that metformin, the most commonly prescribed diabetes medication in the world, appears to exert a meaningful portion of its metabolic benefits through the gut microbiome.
A study by Forslund and colleagues, published in Nature in 2015, found that metformin treatment was associated with significant shifts in gut microbial composition, including an increase in Akkermansia muciniphila and other bacteria associated with metabolic benefit. This suggests that the gut microbiome is not a passive bystander in diabetes treatment but an active target.
Diet Shapes the Microbiome, and the Microbiome Shapes Blood Sugar
The gut connection has immediate practical relevance. Diet is the most powerful tool for modifying gut microbial composition. A diet rich in diverse plant foods and dietary fibre supports the growth of butyrate-producing bacteria and other beneficial species.
A diet high in ultra-processed foods, refined carbohydrates, and saturated fat does the opposite: it depletes microbial diversity and promotes the growth of bacteria associated with inflammation and metabolic disruption.
This creates a reinforcing loop in both directions. Better dietary choices improve the gut microbiome. An improved gut microbiome supports better blood sugar control and reduces inflammation. That, in turn, makes further lifestyle changes more sustainable.
Fermented foods such as yoghurt, buttermilk, kefir, kimchi, kombucha, and sauerkraut introduce live beneficial microorganisms into the gut and may support microbial diversity.
Prebiotic-rich foods, including garlic, onions, leeks, Jerusalem artichokes, and legumes, feed existing beneficial bacteria. Research in this area is evolving rapidly, and while targeted probiotic and prebiotic supplementation remains an area of active investigation, dietary approaches to gut health are already well-supported by evidence.
Putting It All Together
Type 2 diabetes is complex, but its core message is clear.
It is a condition driven by insulin resistance and progressive beta cell dysfunction, shaped by genetics, lifestyle, body composition, and increasingly, the state of the gut microbiome.
It can lead to serious complications if left unmanaged: cardiovascular disease, kidney failure, blindness, and nerve damage. But it is also extraordinarily responsive to the choices made every day.
Whether you are managing a diagnosis, supporting someone who is, or looking to reduce your own risk, the takeaway is the same: small, sustained changes in how you live make a profound difference. And the earlier those changes begin, the greater the benefit.
FAQs
Yes. If unmanaged, type 2 diabetes can damage the heart, kidneys, nerves, and eyes. Early diagnosis and healthy lifestyle changes can help control blood sugar and reduce long-term complications.
Not always. Many people manage type 2 diabetes with diet, exercise, and medication. Insulin may be needed later if the body cannot produce enough insulin naturally.
People with obesity, a family history of diabetes, inactive lifestyles, PCOS, high stress, or poor eating habits are at higher risk of developing type 2 diabetes.
Yes, genetics can increase the risk of type 2 diabetes. However, lifestyle factors like diet, exercise, sleep, and stress also play a major role in its development.
Fasting may help improve insulin sensitivity in some people, but it is not suitable for everyone. People with diabetes should consult a doctor before trying fasting routines.
Disclaimer: This article is for educational and informational purposes only. It is not a substitute for professional medical advice, diagnosis, or treatment. Always consult a qualified healthcare provider before making changes to your diet, medication regimen, or lifestyle.
References
- Sun H, et al. IDF Diabetes Atlas: Global, regional and country-level diabetes prevalence estimates for 2021 and projections for 2045. Diabetes Research and Clinical Practice. 2022. https://pubmed.ncbi.nlm.nih.gov/34879977/
- Knowler WC, et al. Reduction in the incidence of type 2 diabetes with lifestyle intervention or metformin. New England Journal of Medicine. 2002. https://www.nejm.org/doi/full/10.1056/NEJMoa012512
- Zinman B, et al. Empagliflozin, cardiovascular outcomes, and mortality in type 2 diabetes (EMPA-REG OUTCOME). New England Journal of Medicine. 2015. https://www.nejm.org/doi/full/10.1056/NEJMoa1504720
- Marso SP, et al. Liraglutide and cardiovascular outcomes in type 2 diabetes (LEADER). New England Journal of Medicine. 2016. https://www.nejm.org/doi/full/10.1056/NEJMoa1603827
- Lean ME, et al. Primary care-led weight management for remission of type 2 diabetes (DiRECT). The Lancet. 2018. https://www.thelancet.com/journals/lancet/article/PIIS0140-6736(17)33102-1/fulltext
- Estruch R, et al. Primary prevention of cardiovascular disease with a Mediterranean diet (PREDIMED). New England Journal of Medicine. 2013. https://www.nejm.org/doi/full/10.1056/NEJMoa1200303
- Reynolds A, et al. Carbohydrate quality and human health: a series of systematic reviews and meta-analyses. The Lancet. 2019. https://www.thelancet.com/journals/lancet/article/PIIS0140-6736(18)31809-9/fulltext
- Sigal RJ, et al. Effects of aerobic training, resistance training, or both on glycemic control in type 2 diabetes. Annals of Internal Medicine. 2007. https://www.acpjournals.org/doi/10.7326/0003-4819-147-6-200709180-00005
- van Son J, et al. Mindfulness-based cognitive therapy for people with diabetes and emotional problems: long-term follow-up findings from the DiaMind randomized controlled trial. Diabetes Research and Clinical Practice. 2018. https://www.sciencedirect.com/science/article/abs/pii/S0168822717302541
- Qin J, et al. A metagenome-wide association study of gut microbiota in type 2 diabetes. Nature. 2012. https://www.nature.com/articles/nature11450
- Karlsson FH, et al. Gut metagenome in European women with normal, impaired and diabetic glucose control. Nature. 2013. https://www.nature.com/articles/nature12198
- Depommier C, et al. Supplementation with Akkermansia muciniphila in overweight and obese human volunteers. Nature Medicine. 2019. https://www.nature.com/articles/s41591-019-0495-2
- Forslund K, et al. Disentangling type 2 diabetes and metformin treatment signatures in the human gut microbiota. Nature. 2015. https://www.nature.com/articles/nature15766





Post Comment